
Engineering On-Body Drug Delivery Systems (OBDS) for High-Viscosity Biologics
Precision springs and metal components enabling reliable large-volume subcutaneous drug delivery
16/03/2026
By Iker Ibisate, Product Engineer at RPK Technological Center and RPK Medical
Biologic therapies, such as monoclonal antibodies and other complex proteins, are now used more widely to treat autoimmune diseases, cancer, and inflammatory conditions. At the same time, healthcare is shifting toward patient-centered models that allow people to receive treatment outside clinics.
This transition is accelerating the development of On-Body Drug Delivery Systems (OBDS), also known as wearable injectors or patch injectors. These devices allow patients to receive large doses of biologic therapies through controlled subcutaneous delivery while the device remains adhered to the body.
However, delivering biologics in wearable devices introduces significant engineering challenges. High-concentration biologics often exhibit high viscosity, large dose volumes, and long delivery durations, placing demanding requirements on device mechanics, fluid control, and reliability.
At RPK Medical, we help develop next-generation drug-delivery devices by designing and manufacturing precision springs and metal parts essential to autoinjectors and wearable injection systems.
The Rise of High-Viscosity Biologics
Many modern biologic drugs are formulated at high concentrations to reduce how often patients need injections and keep injection volumes smaller. Monoclonal antibodies, for example, can reach over 100 mg/mL, making the fluid much thicker.
Historically, subcutaneous injections were limited to 1–2 mL per injection site, with some applications extending toward 3 mL. When therapies require larger doses, developers must either increase drug concentration or use large-volume subcutaneous delivery technologies.
As a result, modern drug delivery systems increasingly need to handle:
- viscosities exceeding 100 centipoise (cP)
- injection volumes between 5 mL and 20 mL
- delivery durations ranging from several minutes to half an hour
These requirements are too much for standard handheld autoinjectors, so more developers are turning to wearable injection systems built for large-volume delivery.
What Are On-Body Drug Delivery Systems (OBDS)?
On-Body Drug Delivery Systems are wearable devices that deliver medication under the skin over a set period while staying attached to the patient.
These devices are used when the drug volume is too high for handheld injectors.
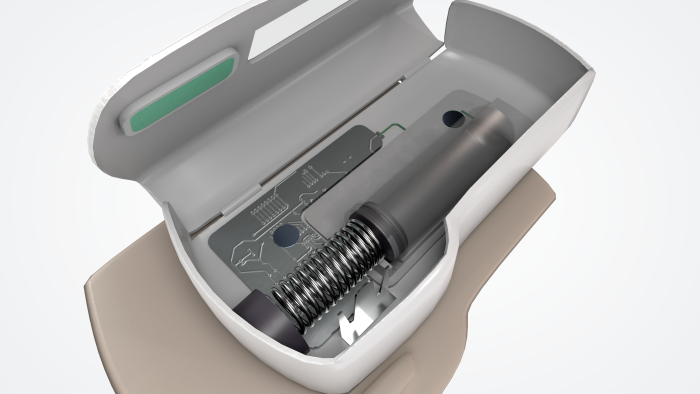
A typical OBDS has several built-in subsystems.
Drug reservoir or container
Most systems use a prefilled syringe or cartridge integrated into a disposable wearable module.
Drive mechanism
Drug delivery is powered by a spring-driven, gas-driven, or electromechanical actuator that generates the force required to move the plunger or pump mechanism.
Needle or soft cannula insertion
An automated mechanism deploys the needle or cannula before controlled drug delivery begins.
Sensors and control systems
Many devices incorporate electronic monitoring systems to detect occlusions, confirm dose completion, and guide user interaction.
Adhesive interface
A medical-grade adhesive securely attaches the device to the skin throughout the delivery period.
Since OBDS devices stay on the patient during daily life, their design needs to balance mechanical performance, comfort, and reliability.
Engineering Challenges in High-Viscosity Drug Delivery
Designing wearable injectors for biologics takes a systems engineering approach. Fluid dynamics, mechanical design, usability, and manufacturing limits all affect each other. OBDS development comes with several technical challenges.
Injection Force and Pressure Management
Highly viscous drug formulations increase resistance in the fluid pathway, particularly when the drug must pass through narrow needles or cannulas.
At the same time, tissue backpressure under the skin changes during the injection.
So the device has to generate enough pressure to maintain the right flow rate without making it bigger or less comfortable for the patient.
This requirement directly influences the design of:
- spring systems
- plunger drive mechanisms
- internal fluid paths
Flow Control and Dose Precision
For large-volume injections, just delivering the right total dose is not enough. The flow rate over time, or dose delivery profile, needs to stay steady throughout the injection.
If force decreases during the stroke or mechanical variability occurs, the device may experience:
- unstable injection speed
- extended delivery time
- incomplete dosing
Keeping mechanical force consistent is a key design goal.
Drug Stability Under Mechanical Stress
Many biologic drugs do not handle mechanical stress well.
High shear rates, abrupt pressure changes, or interactions with internal surfaces can contribute to protein aggregation or particle formation.
For this reason, the design of fluid pathways and wetted components must minimize:
- high-shear restrictions
- sudden pressure spikes
- air entrainment
Choosing the right materials and surface finish is essential here.
Wearability and Adhesion
Unlike handheld devices, OBDS platforms have to stay attached to the patient during daily activities.
Engineering considerations include:
- device weight and center of gravity
- adhesive surface area and durability
- stable needle or cannula placement
How wearable the device is affects both how patients feel and how reliably they get their dose.
Hardware Architectures: From Autoinjectors to Wearable Injectors
Drug delivery platforms are changing because biologic therapies now need larger volumes and higher viscosities.
Traditional autoinjectors remain common for small-volume injections, whereas wearable injectors are used for larger doses.
Feature | Large-Volume Autoinjector | On-Body Drug Delivery System |
Typical volume | 2–5 mL | 5–50 mL |
Delivery time | 10–20 seconds | 5–30 minutes |
Drive system | Compression spring | Constant-force spring or motor |
Patient interaction | Handheld | Adhered to body |
Wearable injectors lower the needed injection force by delivering medication more slowly and at a lower flow rate.

Precision Spring Engineering in Drug Delivery Devices
Springs are fundamental mechanical components in many injection devices.
How well they work affects force generation, flow stability, and dose accuracy.
Compression Springs in Autoinjectors
Traditional autoinjectors usually use high-force compression springs to deliver the dose quickly.
These springs are commonly manufactured from 17-7PH stainless steel, a material known for high strength and fatigue resistance.
However, compression springs follow Hooke’s law, meaning the force decreases as the spring expands. In high-viscosity applications, this can slow injection speed toward the end of the dose.
Constant-Force Springs for Wearable Injectors
Wearable injection systems often use constant-force springs that keep the same force throughout the delivery.
This allows the device to maintain:
- consistent flow rate
- controlled dose delivery profile
- stable injection performance
These features matter most when delivering large volumes of thick biologics.
Multistage Spring Mechanisms
In some cases, the device has to overcome both the initial friction of the stopper and the resistance from the fluid.
Multistage or telescopic spring systems can provide:
- high initial break-loose force
- lower sustained glide force
This approach optimizes mechanical energy distribution while protecting the device housing and primary container.
The Role of Precision Metal Components in OBDS
Besides springs, wearable drug delivery systems use many precision metal parts that support both mechanical and electromechanical functions.
Examples include:
- compression and constant-force springs
- electrical contact elements
- stamped and formed metal parts
- locking and safety mechanisms
- actuator engagement systems
Manufacturing these components requires tight dimensional tolerances, controlled materials, and scalable production processes.
At RPK Medical, we use spring engineering, precision stamping, and metal forming to make parts for advanced drug-delivery devices. Our engineers work closely with device developers to help with manufacturability, reliability, and system integration.
Engineering the Future of Wearable Drug Delivery
The next generation of drug delivery systems is moving toward:
- connected wearable devices
- sensor-enabled delivery systems
- adaptive flow control mechanisms
- hybrid mechanical and electromechanical architectures
Even with these advances, precision mechanical parts are still at the heart of device performance.
Reliable springs and small metal mechanisms make sure the device delivers consistent force, controlled drug delivery, and long-term reliability.
For companies developing wearable injectors, working early with specialized engineering partners helps solve key design challenges like force generation, miniaturization, manufacturability, and reliability.
At RPK Medical, we support these innovations by engineering and manufacturing precision springs and metal components for advanced drug delivery devices.

Frequently Asked Questions
What is an on-body drug delivery system?
An on-body drug delivery system is a wearable injector that adheres to the patient’s body and delivers a medication subcutaneously from an integrated reservoir or cartridge over a controlled period.
Why are high-viscosity biologics difficult to inject?
High-viscosity biologics create greater flow resistance during injection. This requires higher injection force and more precise control of flow rate, which increases the engineering complexity of drug delivery devices.
What is the difference between an autoinjector and a wearable injector?
Autoinjectors are handheld devices typically used for small-volume injections delivered within seconds. Wearable injectors, such as OBDS devices, adhere to the body and deliver larger volumes over several minutes.
Why are constant-force springs used in wearable injectors?
Constant-force springs maintain a stable force throughout the entire delivery stroke. This helps ensure consistent flow rate, controlled injection performance, and reliable dose delivery.
What metal components are used in drug delivery devices?
Drug delivery devices often incorporate metal components such as springs, electrical contacts, stamped parts, locking mechanisms, and actuator elements that support the device’s mechanical and electromechanical functions.
How does RPK Medical support drug delivery device manufacturers?
RPK Medical supports device developers by engineering and manufacturing precision springs, stamped metal parts, and bent components for drug delivery devices, helping ensure reliable performance and scalable manufacturing.
Let’s talk and turn concerns into certainties. Tell us!
Get in touch with our engineering and sales teams, we’ll listen to you carefully and find the best solution together.
Contact us now